Investigating Ocean Acidification using Shells and Calcium Carbonate
|
Introduction
The oceans are a vital part of the Earth, not only is it an ecosystem that supports various life forms, it covers 71% of the Earth's surface. Birds, bears, and other land mammals rely on oceanic animals for sustenance. Many food chains are also interwoven with species only found in the ocean. Important primary producers such as phytoplankton and zooplankton thrive in aquatic environments such as the ocean. The oceans have also provided many poets, authors, artists, and others with deep metaphorical meanings to include in their work. Many people rely on the ocean for food such as fish and shrimp, or for other types of professions. The ocean is so important to the world, and yet it is abused by mankind through: dumping of waste, dumping of hazardous wastes, overfishing, and continuously rising carbon dioxide levels. Each of these problems lead to the devastation of ocean ecosystems that the entire world relies on. The dumping of garbage had led to a decrease in sustainability because the trash that invades ecosystems often end up as lunch for unsuspecting sea animals such as turtles who often mistake plastic trash bags for jellyfish and then end up choking on the trash bag. Chemicals such as Mercury are also dumped into the ocean from factories of cargo ships. These chemicals are released by, "coal-fired power plants, boilers, steel production, incinerators, and cement plants." (NRDC). Humans have also depleted biodiversity in the oceans by overfishing. According to the World Wildlife Fund, often targets big predators such as sharks, as these secondary consumers are wiped from the food chain, large populations of small primary consumers populations increase due to a lack of predators. If mollusk shells are placed in a solution with a low pH, then the shell will be weakened and break easily. If mollusk shells are placed in a solution that is slightly basic (a pH of about 7, which is the pH of salt water), then the shell will remain the same. |
Methods and Materials
Two untreated shells were removed from their bag and labeled "C" (control) and "E" (experimental). The initial qualitative information of both shells were recorded. Then the mass was found for each of the shells and recorded. 150 ml of vinegar were poured into a 500mL beaker and 150 mLs of salt water was poured into a separate beaker. The pH of the substances was tested using pH strips. The color the pH strip turned was recorded. At the same time, the untreated "C" shell was added to the salt water and the untreated "E" shell was added to the vinegar. Once in the solutions a timer was set for 30 minutes. The group then observed the shells and recorded the state of the shells at 0 minutes, 15 minutes, and 30 minutes. In between, the mass of the pre-treated high exposure and low exposure shells was found. once the thirty minutes were over, the untreated shells were removed from their solutions and placed on a paper towel. The shells were dried then massed again.
The strength of the high exposure shells and low exposure shells was tested by placing a biology textbook on the shells and slowly adding more textbooks until the shells broke.
Results
Shells Initial Mass Final Mass Difference Observations
_________________________________________________________________________________________________________________________
Control (C) 2.235 g 2.350 g -0.085 g 7 books
_________________________________________________________________________________________________________________________
Experimental (E) 1.810 g 1.790 g 0.020 g 6 books
_________________________________________________________________________________________________________________________
Low (L) 2.010 g 2.000 g 0.010 g 2 books
_________________________________________________________________________________________________________________________
High (H) 2.000 g 1.978 g -0.022 g 3 books
_________________________________________________________________________________________________________________________
The data shows that the lower the pH a shell was exposed to, its mass decreased. It also shows that shells in salt water were stronger than those exposed to an acidic environment.
Conclusion
The data supports the hypothesis because the shells that were exposed to vinegar, which is acidic, were weaker and had lost some of its mass. This investigation is relevant to the environment because as the ocean becomes more acidic, organisms that do not have the adaptation of a hard shell will be affected negatively because they will have no protection against the lowering pH of the water. Shelled organisms themselves are not safe either, they only have a slight advantage over other organisms. This affects societies because many jobs that rely on fishing or organisms within the oceans will be disadvantaged due to smaller population sizes.
Investigating ocean acidification by changing the ph of salt water
|
Introduction
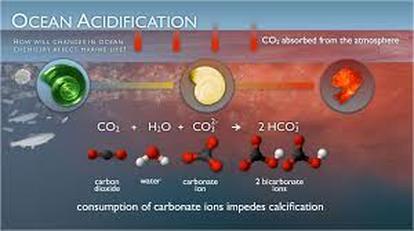
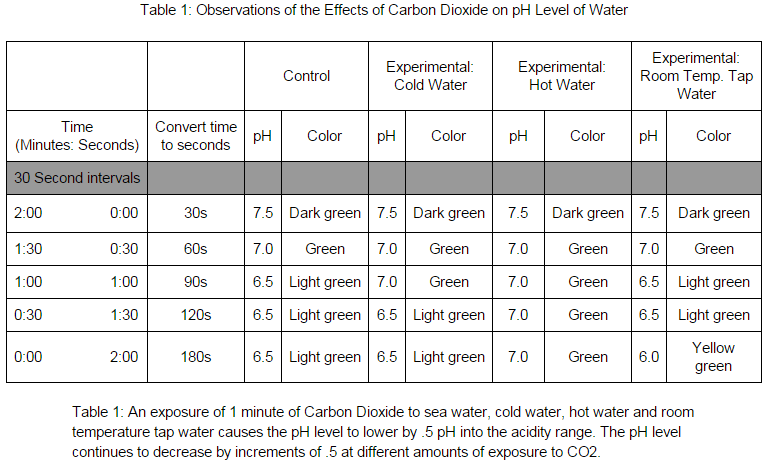
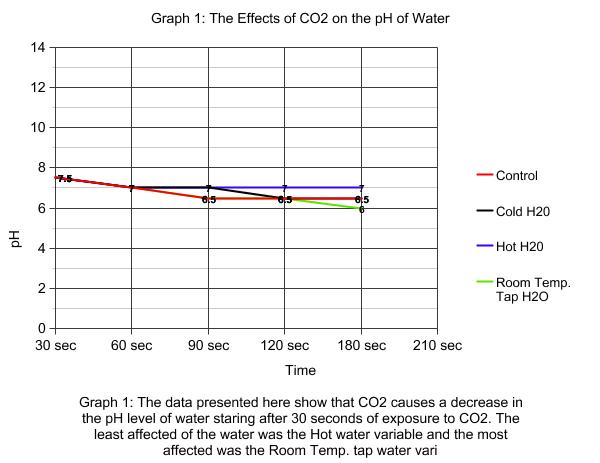
Carbon dioxide emissions are known to be one of the main causes of Global Warming which in turn has led to the melting of the polar ice caps. The warming of water and the climate is also attributed to Global Warming. However, Global Warming is not the only outcome of uncontrolled carbon dioxide emissions. Ocean acidification is an outcome that is devastating all of the ecosystems that the ocean supports. "When carbon dioxide (CO2) is absorbed by seawater, chemical reactions occur that reduce seawater pH, carbonate ion concentration, and saturation states of biologically important calcium carbonate minerals." (PMEL) In simpler terms the higher the levels of carbon dioxide are in the atmosphere, the ocean will take in more carbon dioxide in order for equilibrium to be reached. As the ocean takes in more carbon dioxide that is converted to calcium carbonate, the pH of the ocean becomes more acidic because it is not being released back into the atmosphere. Although there are many life forms in the ocean that require calcium carbonate to compose their shells and backbones, there are not enough of them to take up all of the calcium carbonate being released. If carbon carbonate levels are increased in a beaker of salt water then the salt water will become more acidic. Methods and Materials Salt water was made by mixing salt in a two liter bottle of water. 100 mLs of the salt water were transferred into a 500 mL beaker with a graduated cylinder. 1 mL of universal indicator was added to the salt water in the beaker. The beaker was placed atop a sheet of white paper to make it easier to observe color changes in the water. The beaker was covered with plastic wrap and a hole was poke through it with a straw. There were three roles assigned to the groups: the timer, recorder, and the breather. The breather would breathe continuously into the solution through the straw for 2 minutes. The recorder would call out thirty second intervals so that the recorder could determine the pH of the water. One of the experimental groups changed the salt water for tap water, but followed the same protocol. The second experimental group had water that was 10 degrees lower than the salt water at room temperature, this was done by placing a beaker of 100 mLs of salt water in an ice bath until the temperature went down 10 degrees. The third experimental group used hot salt water. 100 mLs of salt water was added to a beaker which was placed on a hot plate, a thermometer was used to determine when the salt water had risen 10 degrees. This followed the same protocol. |
Results
Conclusion
The data supports the hypothesis because as more calcium carbonate was breathed into the beaker, the acidity levels rose as evidenced by the changing colors of the universal indicator added to the salt water. This investigation can be related to modern society and environments because if humans increase carbon dioxide levels and the levels of other chemicals that have a direct affect on oceanic ecosystems, then life in the oceans will cease to exist. Which will result in the destruction of entire ecosystems and the collapse of many food webs.
The data supports the hypothesis because as more calcium carbonate was breathed into the beaker, the acidity levels rose as evidenced by the changing colors of the universal indicator added to the salt water. This investigation can be related to modern society and environments because if humans increase carbon dioxide levels and the levels of other chemicals that have a direct affect on oceanic ecosystems, then life in the oceans will cease to exist. Which will result in the destruction of entire ecosystems and the collapse of many food webs.